One
of the most important and universally encountered situations in evaluating
in-service corrosion is assessment of influence of oxygen or aeration on
corrosion. Typically, when solutions contain oxygen by virtue of having been
exposed to air, the severity of corrosion increases. Furthermore, under aerated
conditions, the likelihood of localized corrosion (e.g. pitting, crevice attack
and stress corrosion cracking) can also dramatically increase with increased
levels of oxygen in the environment.
In
general terms, aeration accelerates anodic corrosion processes. This is why
both general and localized corrosion are stimulated by aeration. Unfortunately,
most engineers do not realize the major influence of relatively low levels of
oxygen contamination. As shown by the data in the figure below, an increase in
dissolved oxygen concentration in seawater from 10 to 100 ppb increases the
general corrosion rate for steel by a factor of ten. Furthermore, the actual
corrosion damage can be even worse since with increased oxygen content,
localized corrosion can increase the rate of attack by another order of
magnitude which can result in an overall 100 times increase in penetration
rate. This increase in corrosivity is dramatic especially since levels of
oxygen saturation of dilute aqueous solutions exposed to air at 24 C and 1 atm
are normally 6 to 8 ppm (4,000 to 8,000 ppb). Typically, the best rule of thumb
is to anticipate aerated conditions unless steps have been taken to exclude air
leakage and to deaerate by mechanical and/or chemical means.
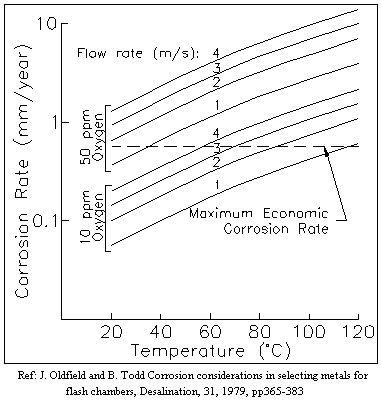
Figure 1. Effect of oxygen concentration as a function of temperature on
corrosion